RECON®. Moving means life. We lead the way.
By combining molecular biology and innovative ceramics, boneArtis AG has created an alternative to artificial joints and embedded a novel procedure for rapid bone growth in orthopaedic and dentistry.
The intelligent implants and the Surgical Diamond Technique (SDI®) enable surgeons for the first time to create fully mineralized bone and joint reconstructions in weeks. The novel procedure has won numerous scientific awards and has been nominated by the Bayern Innovative “Top 5 Innovation Life Award" and 2024 four times honored with a Gold Stevie®Award.
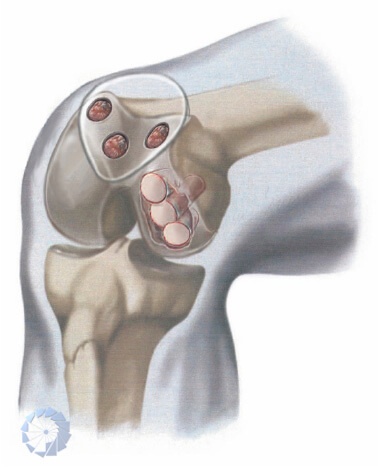
RECON® is the ISO certified academy, helping you as surgeon to resurface cartilage-bone defects and augment bone within weeks.
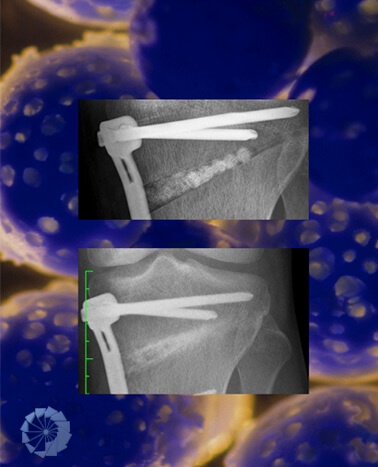
Today, osteoarthritic joints are routinely replaced in a standard procedure with artificial components. For many patients suffering osteoarthritis, large-scale cartilage and bone defects, or trauma, boneArtis is providing with RECON® and the Ceraball® Principle an efficient solution to preserve own joints, by reconstruction and resurfacing, using the SDI®(Surgical Diamond Technique), formerly known as DBCS® (Diamond Bone Cutting System).
The innovative treatment has helped many patients around the word, enabling a new quality of life and is the alternative to artificial joints.
The transformation from being resurfaced by the RECON® prevented an artificial joint has been transformed into a emotional touching documentary, which got awarded 2024 as best documentary at the Stevie®Awards.
Our Services
and Products
SDI – Surgical Diamond Instruments:
- Surgical Diamond Instruments for reconstruction of joints in case of osteoarthritis
- Surgical Diamond Instruments for bone dowelling
- Surgical Diamond Instrumentation for ligament repair
- Surgical Diamond Instrumentation for reconstruction of joints in hopeless cases:
- Very often the chance for high level athletics to return to sports activity
- A chance for children and young adults to save joint function in osteochondritis dissecans
- A chance to treat osteo-cartilage fragments
- A chance to re-unite non-unions in fracture treatment